Groupe ATP-CGPharm is the ideal partner for the life sciences industry!
In this post, we will discuss our expertise in : Environmental Monitoring.
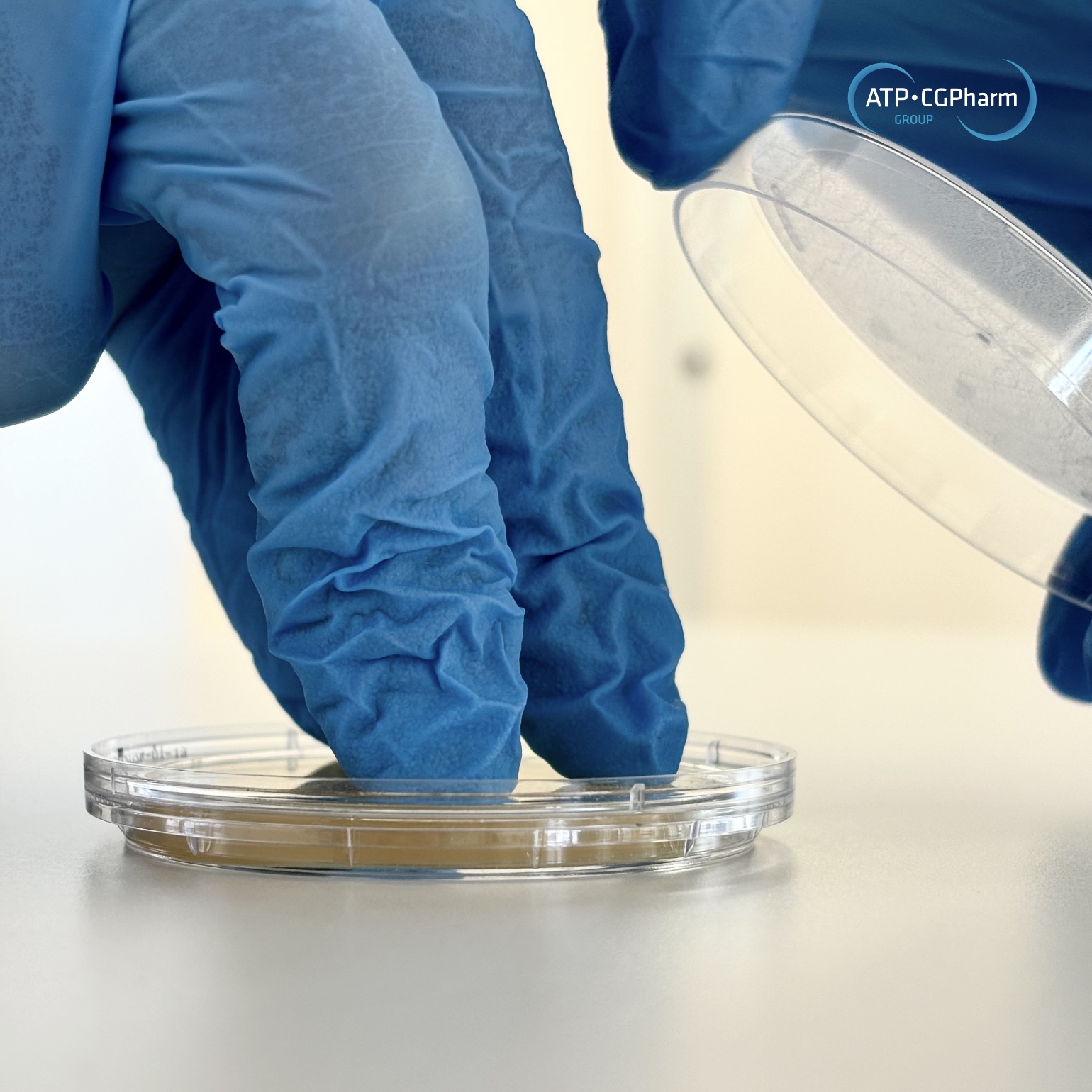
Environmental and utilities qualification and monitoring are crucial challenges faced by the health industries. These requirements are of key importance both from a regulatory point of view and in terms of guaranteeing product quality and process reliability, thereby helping to ensure patient safety by preventing adverse environmental conditions.
Since 1998, the ATP-CGPharm group has had in place a team of trained sampling technicians, qualified and calibrated equipment and sampling materials adapted to the environmental control of health industries.
From simple advice to complete qualification or monitoring of :
Clean rooms (viable particles, non-viable particles, surface samples, etc.)
Purified water, water for injection, pure steam (physico-chemical and microbiological parameters, appearance, etc.)
Compressed air, process gases: O2, CO2, N2, etc. (non-viable particles, physico-chemical and microbiological parameters, dew point, etc.)
ATP-CGPharm is here to help you through every step of the way in compliance with regulation (e.g. GMP, BPF, ISO 14644-1…). From preparing risk analyses, protocols or forms, to carrying out sampling and analytical tests, interpreting results, writing reports and establishing monitoring procedures.
Contact ATP-CGPharm Group for your environmental qualification or monitoring projects!