B
B Company
B SERVICES
For more than 20 years, the ATP-CGPharm Group has been supporting clients in the Life Science Industry with a broad range of services in GxP engineering, commissioning, qualification validation, quality assurance, quality control and environmental monitoring.
B HISTORY
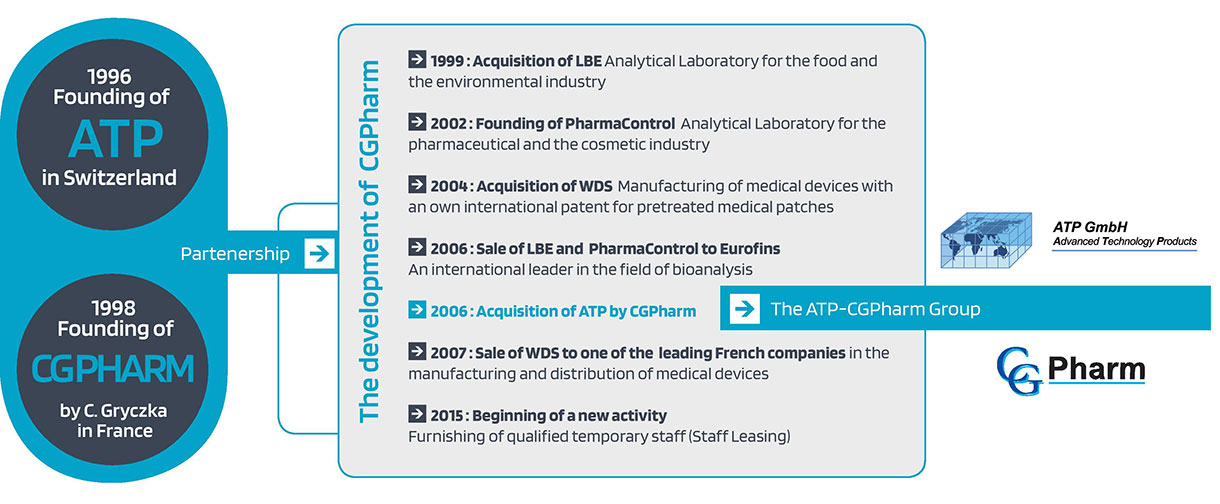
Originally, ATP and CGPharm were two separate companies. ATP was founded in 1996 in Switzerland and CGPharm in 1998 in France.
ATP was experienced in engineering manufacturing plants for pharmaceutical companies while CGPharm was specialized in GMP Compliance consulting for the Life Science Industry.
The two companies quickly came together to conclude a partnership in order to pool their skills and deal with the complexity of the regulatory conditions of the pharmaceutical industry.
At the same time, CGPharm has developed new activities that have enriched its skills and experience:
> In the field of quality control:
– 1999: Acquisition of LBE, a contract analytical laboratory for the food industry and the environment (this company was sold to Eurofins in 2006).
– 2002: Creation of PharmaControl, a contract analytical laboratory for the Pharmaceutical and Cosmetic Industry (this company was sold to Eurofins in 2006).
> In the production of medical devices:
– 2004: Acquisition of WDS, Manufacturer of innovative plasters with its own international patent (this company was sold in 2007).
In 2006, CGPharm acquired ATP to form the current ATP-CGPharm group, a consulting and engineering company serving the Life Science Industry.
B MANAGEMENT
The ATP-CGPharm Group is managed by its owner, Dr. Christophe GRYCZKA, Pharmacist and Consultant for the Pharmaceutical Industry for more than 25 years.
With his own experience in managing companies specializing in quality control and production of medical devices, through his own companies he created, he acts today on behalf of his clients as he would for its own account, constantly seeking the best GxP-conform solutions at the best costs.
Dr. Christophe GRYCZKA is a partner recognized by its customers who appreciate its singularity. Indeed, in addition to his business leadership activities, he continues to be in the field as a project consultant. This particularity allows him to benefit from a great expertise of the GMP and other regulations in all areas of life sciences but also to be fully aware of changes and technological evolutions of the field.

Dr. Christophe GRYCZKA
Pharmacist
Owner CEO
B TEAM
Our team continues to grow by combining the international and multidisciplinary skills of consultants from diverse backgrounds, all oriented towards excellence.
Indeed, our team puts together pharmacists, engineers and technicians from 10 different nationalities who speak English and German fluently and most of them also French.
Our team which is composed of talented young professionnals as well as experienced experts meets regularly to share best practices and its experiences to constantly improve their level of expertise in the projects entrusted to them.
The diversity of our consultants and their commitment enable us to offer a wide range of tailor-made services and customized solutions to our clients in the areas of engineering, qualification validation, quality assurance, quality control and environmental monitoring. For all these skills, we furnish qualified temporary staff with recognized know-how.
All our employees are employed on permanent basis. This as well as a very low staff attrition make it possible to provide lasting support to clients.
B VALUES
The ATP-CGPharm Group is founded on human values that are dear to it by ensuring the satisfaction of its customers and the well-being of its employees.
The success of our projects is based on a motivated team with an open mind to challenges. For this we surround ourselves with committed, passionate employees showing team spirit and willingness to go beyond themselves to support our customers in their daily challenges.
The ATP-CGPharm Group identifies with sporting values, that is why it has been sponsoring SC Freiburg football club for several years.